研究業績
原著論文
- Cationic iridium-catalyzed enantioselective decarboxylative aryl addition of aromatic carboxylic acids to bicyclic alkenes
R. Asano, R. Nonami, H. Hamasaki, K. Kanemoto, H. Fujita, K. Yamamoto, T. Seki, K. Shibatomi, S. Kuwahara, T. Shirai*
Chem. Commun. 2026, in press. DOI: 10.1039/D6CC00389C
【Inside Front Cover】
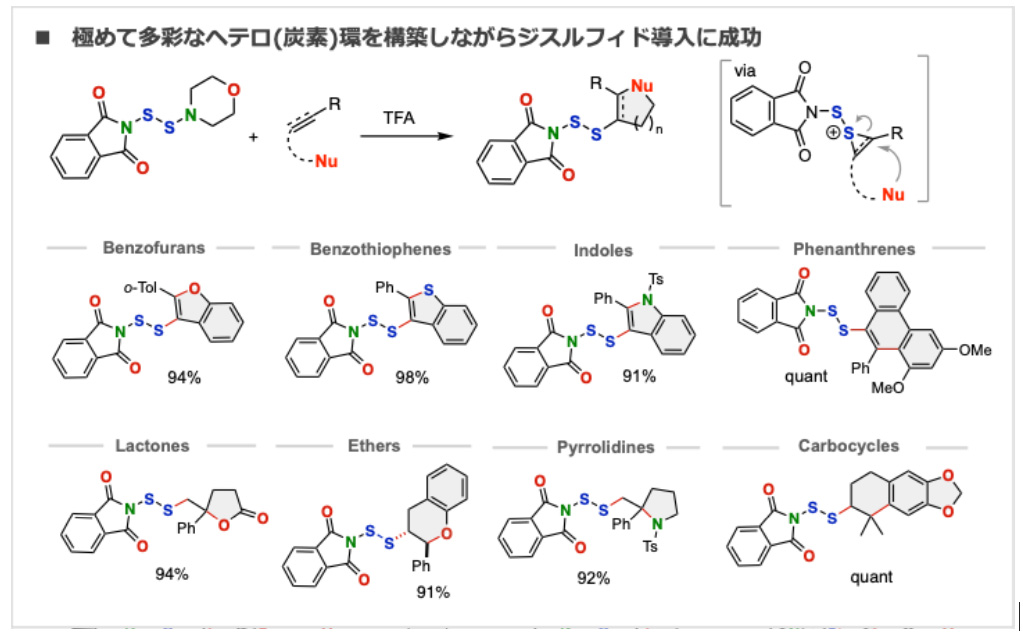
- Dithiofunctionalization: a versatile approach for constructing complex disulfides from alkenes and alkynes
Y. Takami, E. Kwon, K. Kanemoto*
Org. Chem. Front. 2026, 13, 1299. DOI: 10.1039/D5QO01625H
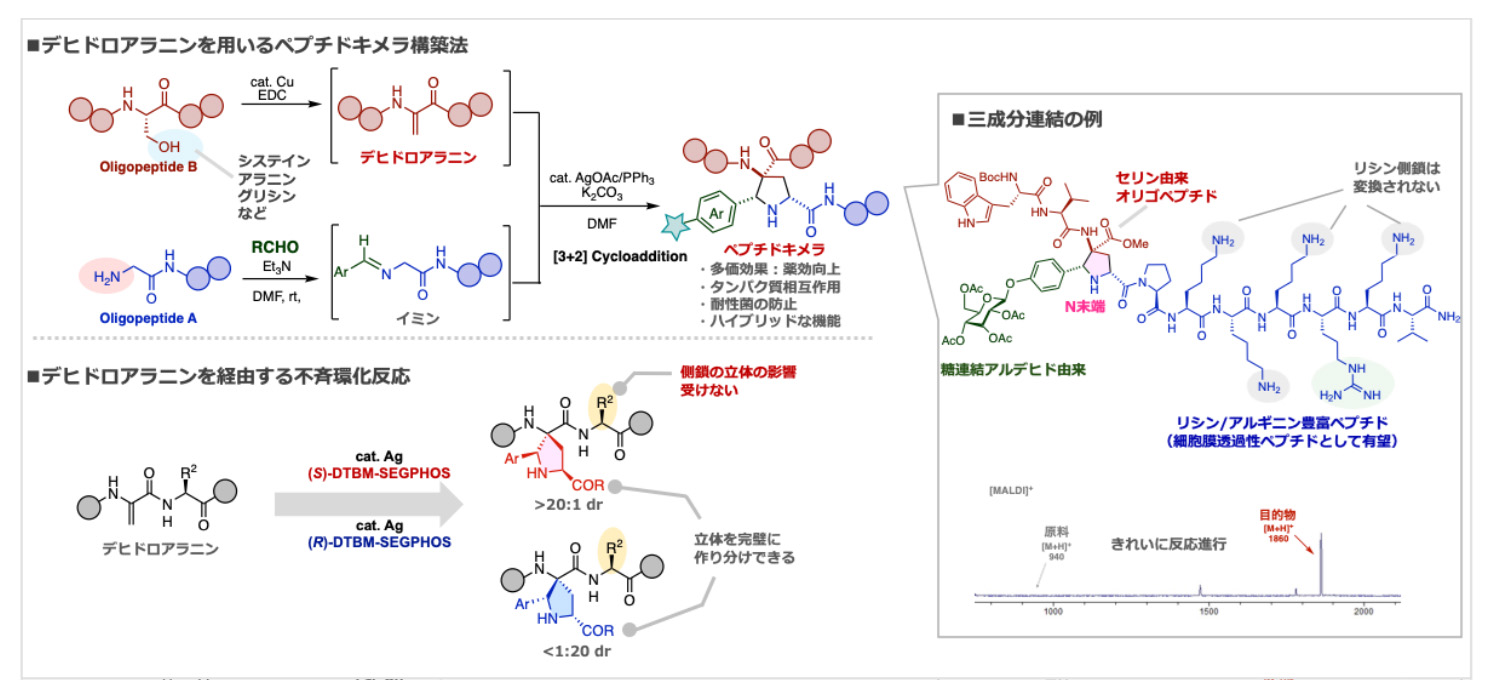
- Precision synthesis of peptide chimeras through site-specific azomethine ylide–dehydroalanine cycloaddition
M. Iwata, Y. Takami, S. Miura, H. Ohno, E. Kwon, N. Yoshikai, K. Kanemoto*
Org. Chem. Front. 2026, 13, 1317. DOI: 10.1039/D5QO01627D
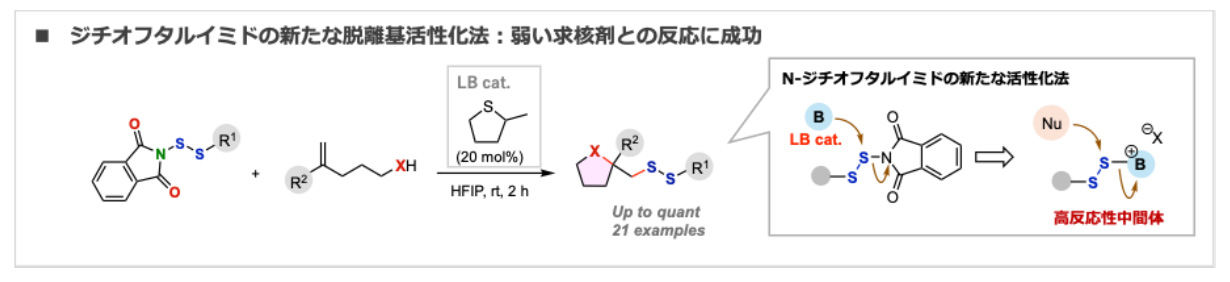
- Preparation of Unsymmetrical Disulfides via Catalytic Lewis Base Activation of N-(Organodithio)phthalimides
H. Ohno, Y. Takami, K. Kanemoto*
J. Org. Chem. 2026, 91, 1817. DOI: 10.1021/acs.joc.5c03056
- Formal Cyclopropylation of Imines with Cyclopropanols: Stereocontrolled Access to Conformationally Constrained γ-Amino Alcohols
K. Tsukiji, K. Kanemoto, E. Kwon, N. Yoshikai*
Angew. Chem. Int. Ed. 2025, 64, e202511646. DOI: 10.1002/anie.202511646
- Hexadehydro Diels–Alder/Alkynyliodanation Cascade: A Highly Regioselective Entry to Polycyclic Aromatics
S. Morohashi, L. Zhou, K. Kanemoto*, E. Kwon, N. Yoshikai*
Org. Lett. 2025, 27, 4269. DOI: 10.1021/acs.orglett.5c00956
- Diazomethyl-λ3-iodane Meets Aryne: Dipolar Cycloaddition and C-to-N Iodane Shift Leading to Indazolyl-λ3-iodanes
S. Otsuki, K. Kanemoto,* D. C. Martos, E. Kwon, J. Wencel-Delord, N. Yoshikai*
Chem. Sci. 2025, 16, 8053. DOI: 10.1039/D5SC00266D
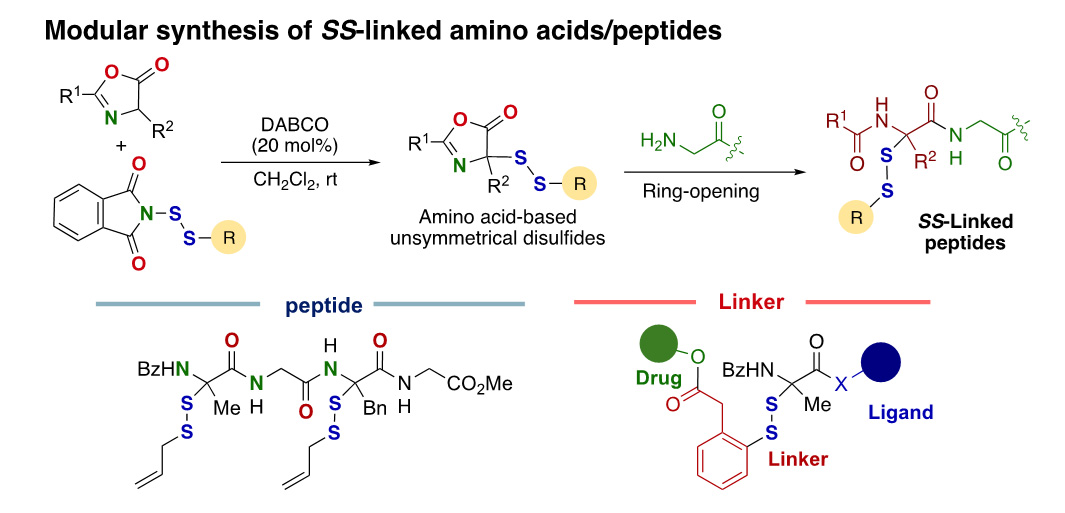
- A versatile entry to unnatural, disulfide-linked amino acids and peptides through the disulfuration of azlactones
M. Iwata, Y. Takami, H. Asanuma, K. Hosono, H. Ohno, N. Yoshikai, K. Kanemoto*
Chem. Sci. 2025, 16, 2777. DOI: 10.1039/D4SC07187E
【プレスリリース】【日経新聞電子版】【日刊工業新聞】【Highlighted in Synfacts, 2025, 21, 429.】
- Stereoselective Hydroxyallylation of Cyclopropenes with Cyclopropanols via NHC Catalysis of Transient Organozinc Species
K. Tsukiji, A. Matsumoto, K. Kanemoto,* N. Yoshikai*
Angew. Chem. Int. Ed. 2024, 63, e202412456. DOI: 10.1002/anie.202412456
【Chem-Station スポットライトリサーチ】
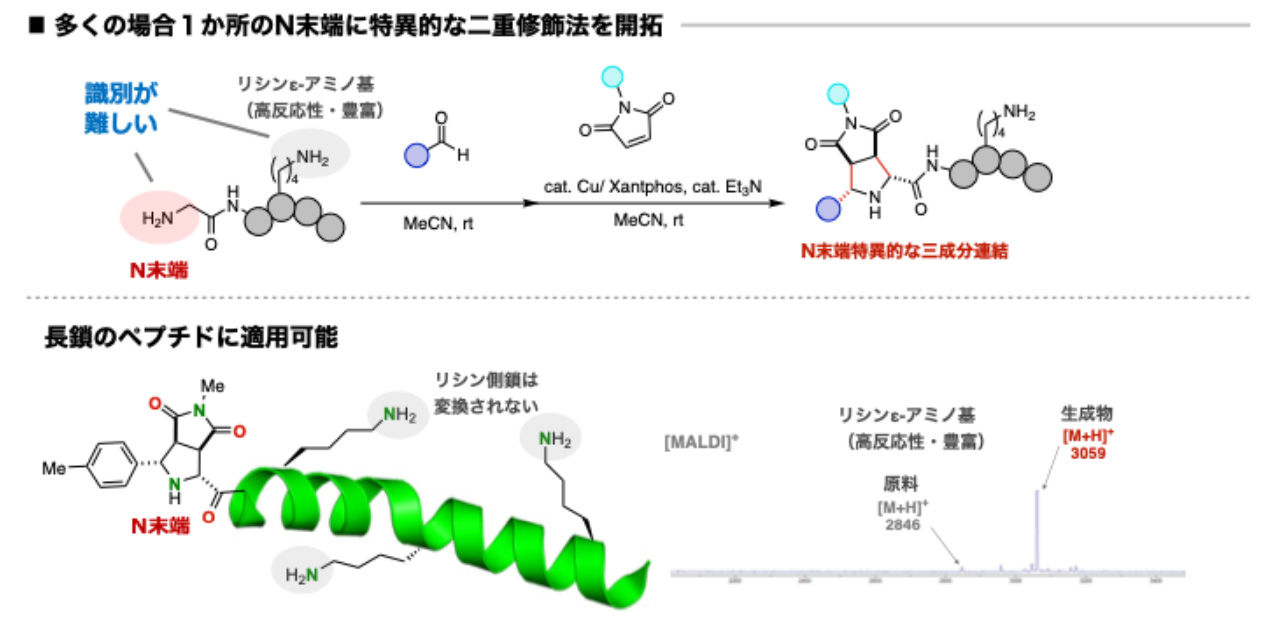
- N‐Terminal‐Specific Dual Modification of Peptides through Copper‐Catalyzed [3+2] Cycloaddition
H. Machida, K. Kanemoto*
Angew. Chem. Int. Ed. 2024, 63,e202320012. DOI: 10.1002/anie.202320012
【プレスリリース】【日経新聞電子版】【Chem-Station スポットライトリサーチ】
- Carboiodanation of Arynes: Organoiodine(III) Compounds as Nucleophilic Organometalloids
C. Arakawa,+ K. Kanemoto,+ K. Nakai, C. Wang, S. Morohashi, E. Kwon, S. Ito, N. Yoshikai*
+Equal contribution.
J. Am. Chem. Soc. 2024, 146, 3910. DOI: 10.1021/jacs.3c11524
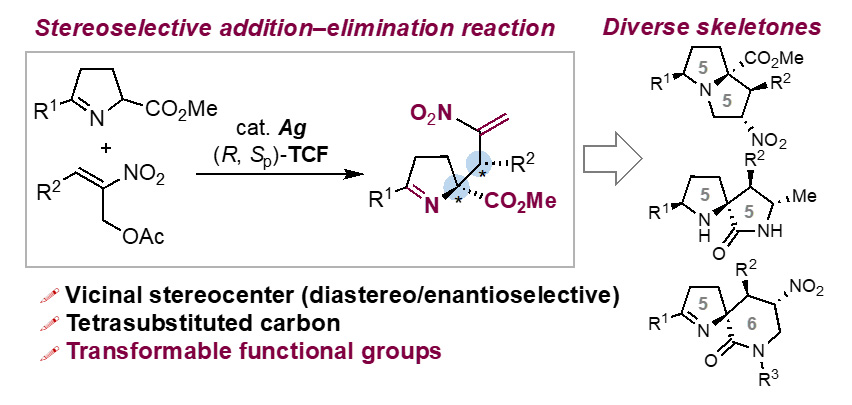
- Construction of Diverse Pyrrolidine-Based Skeletons through Ag-Catalyzed Stereoselective Addition–Elimination Reaction of Azomethine Ylides with Nitroallyl Acetates
I. Ohno, K. Kanemoto*, S. Furuya, Y. Suzuki, S.-i. Fukuzawa*
Org. Lett. 2024, 26, 1880. DOI: 10.1021/acs.orglett.4c00184
- Amino- and Alkoxybenziodoxoles: Facile Preparation and Use as Arynophiles
K. Kanemoto, K. Yoshimura, K. Ono, W. Ding, S. Ito, N. Yoshikai*
Chem. Eur J. 2024, 30, e202400894. DOI: 10.1002/chem.202400894
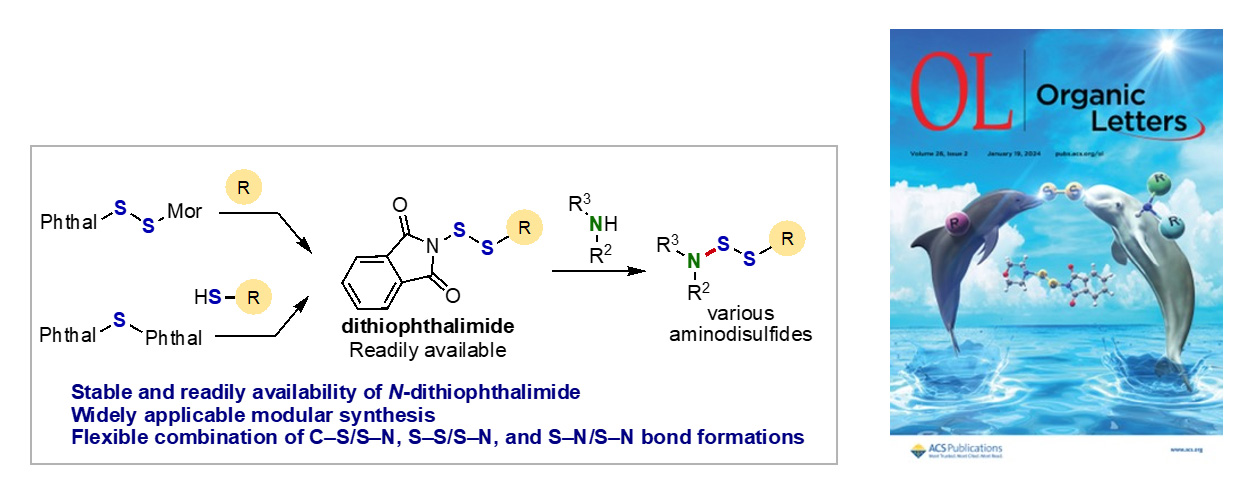
- Amination of N-(Organodithio)phthalimides for the Modular Synthesis of Aminodisulfides
H. Asanuma, K. Kanemoto* Org. Lett. 2024, 26, 438. DOI: 10.1021/acs.orglett.3c03419
【Selected as Front Cover】
- Dipolarophile-Steered Formal Stereodivergent Synthesis of 2,5-cis/trans-Pyrrolidines Based on Asymmetric 1,3-Dipolar Cycloaddition of Imino Lactones
S. Furuya, K. Muroi, K. Kanemoto, S.-i. Fukuzawa*
Chem. Eur. J. 2023, 29, e202302609. DOI: 10.1002/chem.202302609
- Cationic Iridium‐Catalyzed Decarboxylation of Aromatic Carboxylic Acids
R. Nonami, Y. Kishino, T. Yamasaki, K. Kanemoto, K. Iwai, N. Nishiwaki, K. Shibatomi, T. Shirai*
Chem. Asian J. 2023, 18, e202300533. DOI: 10.1002/asia.202300533
- Zinc‐Mediated Diastereoselective Annulation of Cyclopropanols with Alkylidenemalononitriles via Enolized Homoenolate
K. Tsukiji, T. Hayakawa, K. Kanemoto, N. Yoshikai*
Asian J. Org. Chem. 2023, 12, e202300114. DOI: 10.1002/ajoc.202300114
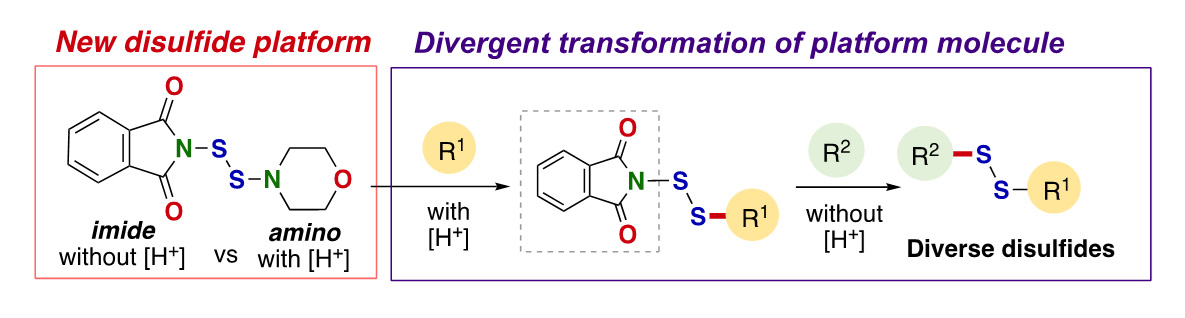
- N-(Morpholine-4-dithio)phthalimide: A Shelf-Stable, Bilateral Platform Molecule Enabling Access to Diverse Unsymmetrical Disulfides
H. Asanuma, K. Kanemoto,* T. Watanabe, S.-i. Fukuzawa*
Angew. Chem., Int. Ed. 2023, 62, e202219156. Angew. Chem. 2023, 135, e202219156. DOI: 10.1002/anie.202219156
【Highlighted in Synfacts, 2023, 19, 0617.】
- Chiral Silver Complex-Catalyzed Asymmetric Conjugate Addition of 1‑Pyrroline-5-Carbonitrile to α‑Enones
H. Araki, S. Furuya, K. Kanemoto, S.-i. Fukuzawa*
J. Org. Chem. 2023, 88, 924. DOI: 10.1021/acs.joc.2c02315
- Diastereoselective Conversion of Cyclopropanols to Cyclopentane-1,3-diols via Aldol Dimerization of Zinc Homoenolates
K. Tsukiji, Y. Sekiguchi, K. Kanemoto, N. Yoshikai*
Chem. Lett. 2022, 51, 1012. DOI: 10.1246/cl.220311
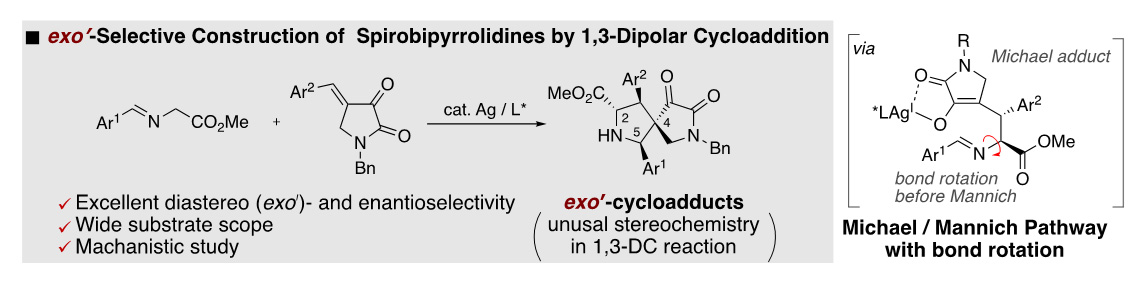
- exoʹ-Selective Construction of Dispiropyrrolidines by the Silver-catalyzed Asymmetric [3+2] Cycloaddition of Imino Esters with 4-Benzylidene-2,3-dioxopyrrolidines
S. Furuya, K. Kanemoto,* S.-i. Fukuzawa*
Chem. Asian J. 2022, 17, e202200239. DOI: 10.1002/asia.202200239
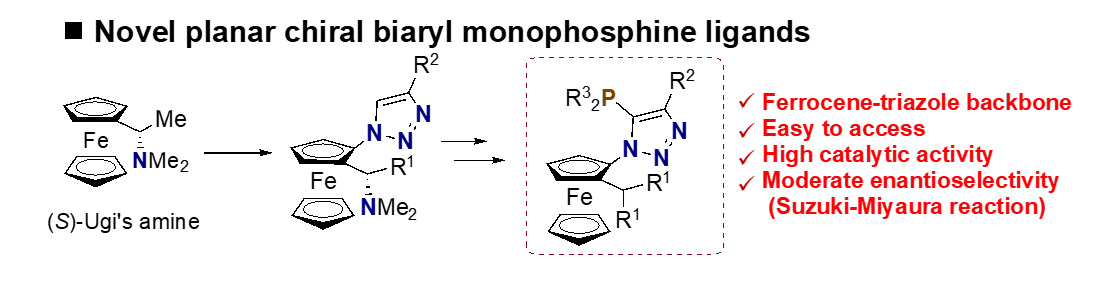
- Synthesis and Evaluation of Novel Planar-Chiral Monophosphine Ligands Bearing Ferrocene-Triazole Backbones
S. Sakai, K. Kanemoto,* S.-i. Fukuzawa*
Eur. J. Inorg. Chem. 2022, e202100967. DOI: 10.1002/ejic.202100967
- Cationic Iridium-Catalyzed Asymmetric Decarbonylative Aryl Addition of Aromatic Aldehydes to Bicyclic Alkenes
R. Nonami, Y. Morimoto, K. Kanemoto, Y. Yamamoto, T. Shirai*
Chem. Eur. J. 2022, 28, e202200317. DOI: 10.1002/chem.202200317
【Selected as supplementary cover】【Selected as hot paper】
- Synthesis and properties of 5,5’-diethynyl indigos and their polymers using Glaser coupling reaction
S. Kenmochi, K. Kanemoto, T. Ikeda, S.-i. Fukuzawa
Fac. Sci. Eng. Chuo Univ. 2022, 27, 35.
- Trifluoroacetic Acid-Mediated Desulfurilative Sulfonylation of Activated Olefins Using Potassium p-Toluenethiosulfonate
T. Watanebe, K. Kanemoto,* S.-i. Fukuzawa
Bull. Fac. Sci. Eng., Chuo Univ. 2022, 27, 15.
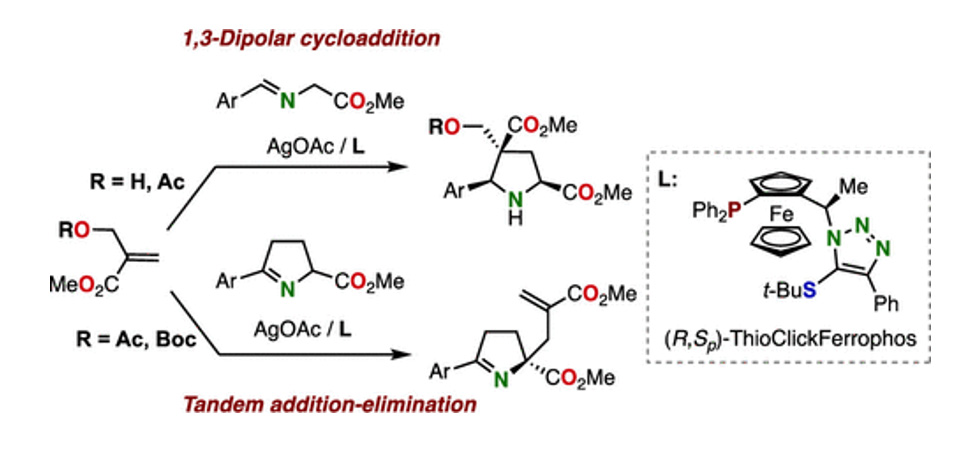
- Silver/ThioClickFerrophos-Catalyzed 1,3-Dipolar Cycloaddition and Tandem Addition-Elimination Reaction of Morita–Baylis–Hillman Adducts
Y. Suzuki, K. Kanemoto,* A. Inoue, K. Imae, S.-i. Fukuzawa*
J. Org. Chem.2021,86, 14586. DOI: 10.1021/acs.joc.1c01440
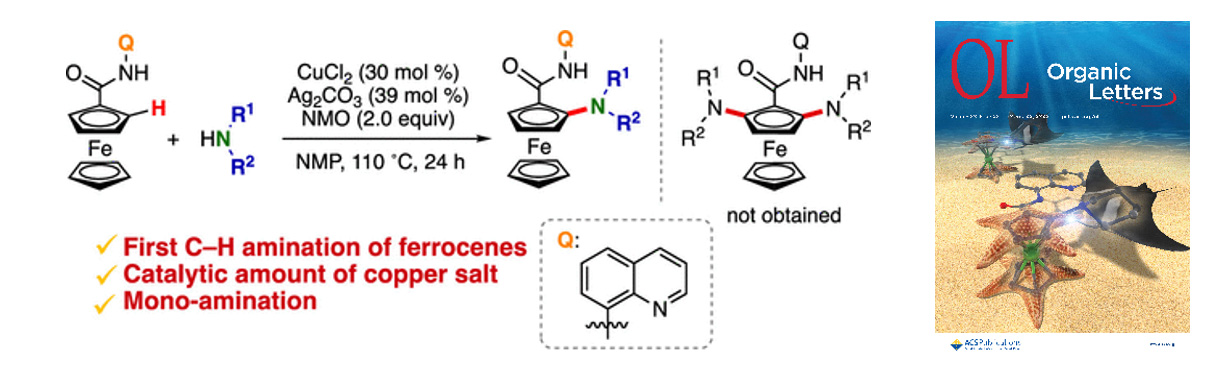
- Copper-Catalyzed Single C–H Amination of 8-Aminoquinoline-Directed Ferrocenes
K. Kanemoto,* N. Horikawa, S. Hoshino, Y. Tokoro, S.-i. Fukuzawa*
Org. Lett. 2021, 23, 4966. DOI: 10.1021/acs.orglett.1c01294
【Selected as supplementary cover】
- Palladium-Catalyzed Sulfinylation of Aryl- and Alkenylborons with Sulfinate Esters
M. Suzuki, K. Kanemoto, Y. Nakamura, T. Hosoya, S. Yoshida*
Org. Lett. 2021, 23, 3793. DOI: 10.1021/acs.orglett.1c01292
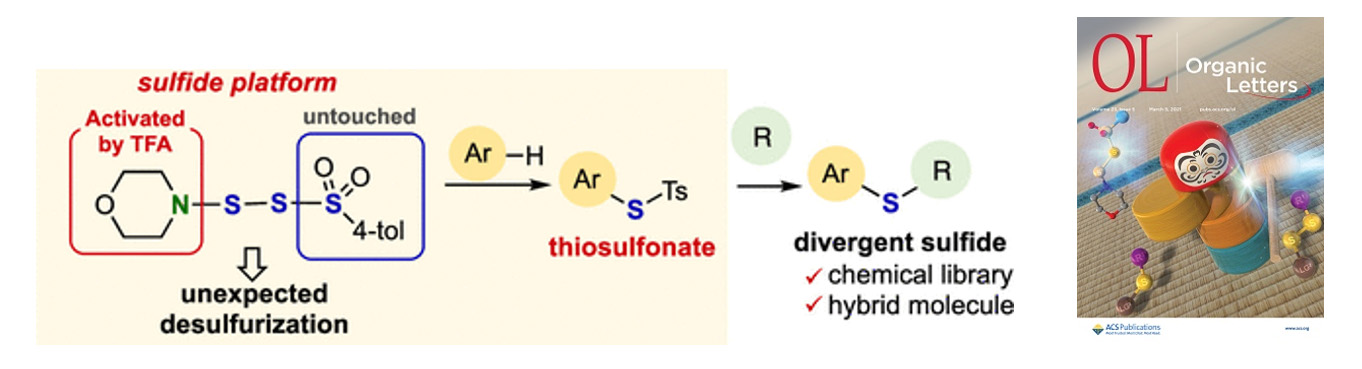
- Acid-Mediated Sulfonylthiolation of Arenes via Selective Activation of SS-Morpholino Dithiosulfonate
K. Kanemoto,* K. Furuhashi, Y. Morita, T. Komatsu, S.-i. Fukuzawa*
Org. Lett. 2021, 23, 1582. DOI: 10.1021/acs.orglett.0c04289
【Highlighted in Organic Chemistry Portal】【Selected as supplementary cover】
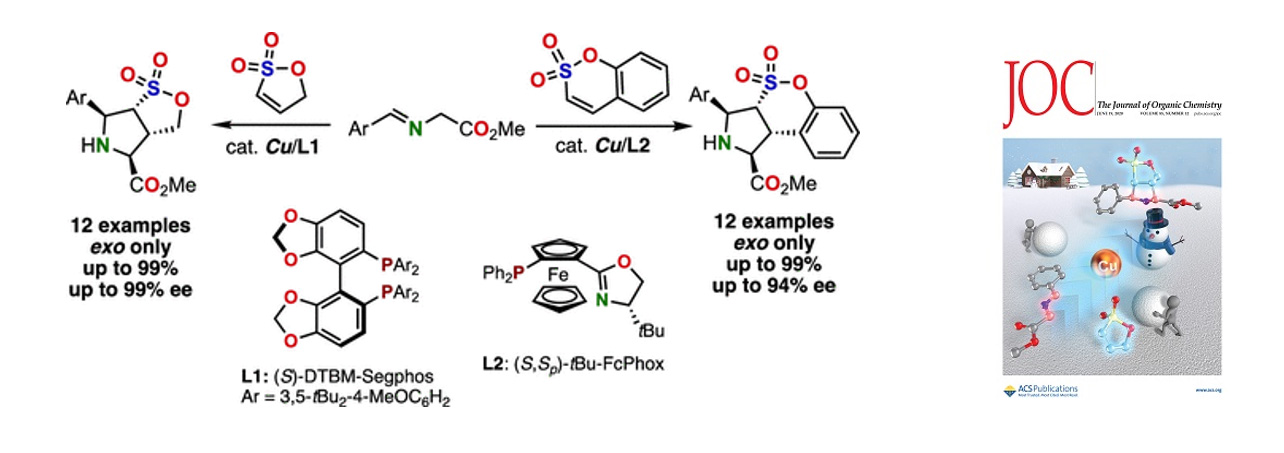
- Copper-Catalyzed Asymmetric 1,3-Dipolar Cycloaddition of Imino Esters to Unsaturated Sultones
S. Furuya, K. Kanemoto,* S.-i. Fukuzawa*
J. Org. Chem. 2020, 85, 8142. DOI: 10.1021/acs.joc.0c01023
【Highlighted in Synfacts, 2020, 10,1118.】
- Cationic Iridium/Chiral Bisphosphine‐Catalyzed Enantioselective Hydroacylation of Ketones
T. Shirai,* T. Iwasaki, K. Kanemoto, Y. Yamamoto
Chem. Asian J. 2020, 15, 1858. DOI: 10.1002/asia.202000386
- Functionalization of a Single C–F Bond of Trifluoromethylarenes Assisted by an ortho-Silyl Group Using a Trityl-Based All-in-One Reagent with Ytterbium Triflate Catalyst
Y. Kim, K. Kanemoto, K. Shimomori, T. Hosoya, S. Yoshida*
Chem. Eur. J. 2020, 26, 6136. DOI: 10.1002/chem.202001315
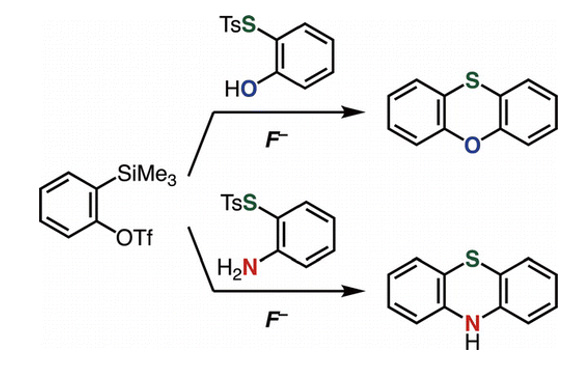
- Synthesis of Phenoxathiins and Phenothiazines by Aryne Reactions with Thiosulfonates
K. Kanemoto, Y. Sakata, T. Hosoya, S. Yoshida*
Chem. Lett. 2020, 49, 593. DOI: 10.1246/cl.200132
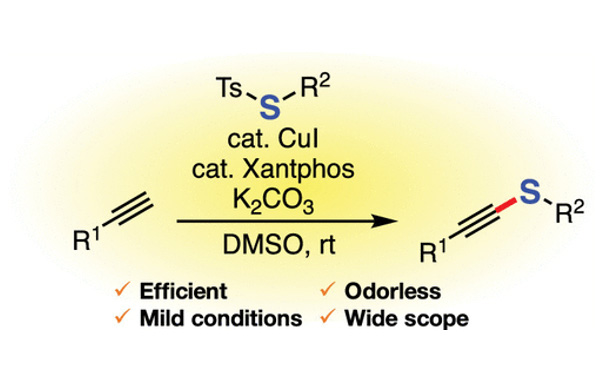
- Synthesis of Alkynyl Sulfides by Copper-Catalyzed Thiolation of Terminal Alkynes Using Thiosulfonates
K. Kanemoto, S. Yoshida*, T. Hosoya*
Org. Lett. 2019, 21, 3172. DOI: 10.1021/acs.orglett.9b00875
【Press Release】【Most downloaded article of Org. Lett. in April 2019】
【Highlighted in Organic Chemistry Portal】
- Copper-Catalyzed Regio- and Diastereoselective 1,3-Dipolar Cycloaddition Reactions of Glycine Imino Esters with 1-Propene-1,3-sultone
S. Furuya, S. Kato, K. Kanemoto, S.-i. Fukuzawa*
Eur. J. Org. Chem. 2019, 4561. DOI: 10.1002/ejoc.201900738
- Facile Synthesis of Diverse o-Iodoaryl Triflates from o-Silylaryl Triflates by Aluminum-mediated Desilyliodination
S. Yoshida*, Y. Hazama, K. Kanemoto, Y. Nakamura, T. Hosoya*
Chem. Lett. 2019, 48, 742. DOI: 10.1246/cl.190223
- Modular Synthesis of Unsymmetrical Doubly-ring-fused Benzene Derivatives Based on a Sequential Ring Construction Strategy Using Oxadiazinones as a Platform Molecule
T. Meguro, S. Chen, K. Kanemoto, S. Yoshida *, T. Hosoya*
Chem. Lett. 2019, 48, 582. DOI: 10.1246/cl.190118
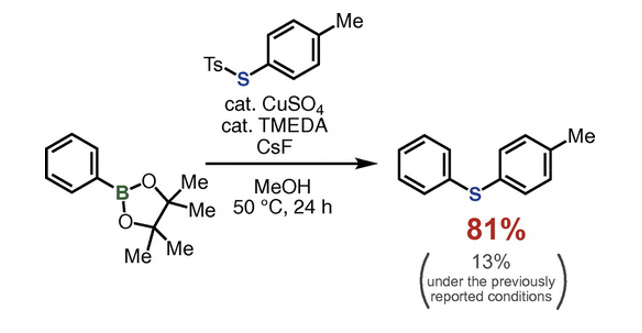
- Modified Conditions for Copper-catalyzed ipso-Thiolation of Arylboronic Acid Esters with Thiosulfonates
K. Kanemoto, S. Yoshida*, T. Hosoya*
Chem. Lett. 2018, 47, 85. DOI: 10.1246/cl.170907
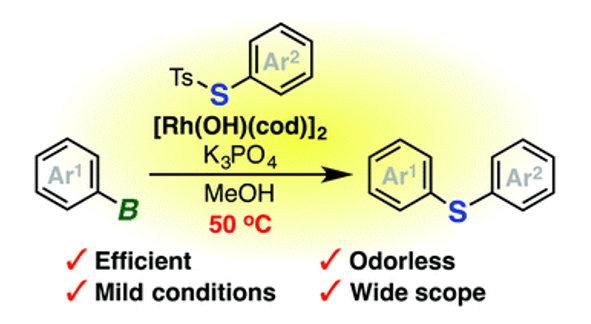
- Rhodium-catalyzed odorless synthesis of diaryl sulfides from borylarenes and S-aryl thiosulfonates
K. Kanemoto, Y. Sugimura, S. Shimizu, S. Yoshida*, T. Hosoya*
Chem. Commun. 2017, 53, 10640. DOI: 10.1039/C7CC05868C